
The year 2009 has just begun, and it looks like it will be a year of big surprises – at least for the makers of Osteoporosis drugs. New information is opening up the door to many unanswered questions and suspicions about the most commonly prescribed drugs such as Fosamax, Actonel, and Boniva.
On December 31st, just on time to herald the New Year, Reuters reported the following:
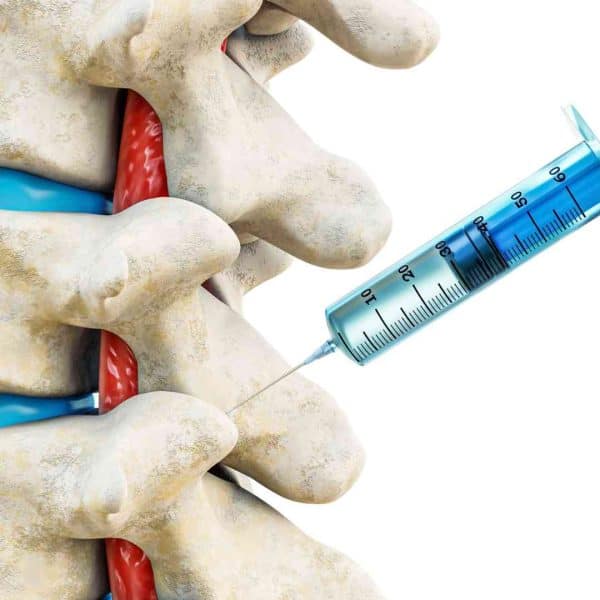
Diane Wysowski of the FDA’s division of drug risk assessment said researchers should check into potential links between bisphosphonate drugs and cancer. In a letter in Thursday’s New England Journal of Medicine, Wysowski said since the initial marketing of Fosamax, known generically as alendronate, in 1995, the FDA has received 23 reports in which patients developed esophageal tumors. Typically, two years lapsed between the start of the drug and the development of esophageal cancer. Eight patients died, she reported. In Europe and Japan, 21 cases involving Fosamax have been logged, with another six instances where Procter & Gamble’s Actonel or risedronate and Didronel or etidronate, and Roche’s Boniva (ibandronate) may have been involved. Six of those people died.
The article also comments on the known fact that these drugs can cause “esophagitis, which is an inflammation of the lining of the tube carrying food to the stomach…which is why patients are instructed to remain upright for at least a half hour after taking them.”
As if this were not enough, on January 2nd 2009 The New England Journal of Medicine
(Volume 360:53-62) published an article titled “Giant Osteoclast Formation and Long-
Term Oral Bisphosphonate Therapy” by Robert M. Weinstein, M.D., et al., that seems to shatter everything we were told about how these drugs work.
Until this study, bisphosphonates were believed to destroy osteoclasts, the crucial bone cells that remove bone so new bone can be deposited. But now – and I’m quoting from the article’s conclusion – “long-term alendronate treatment is associated with an increase in the number of osteoclasts, which include distinctive giant, hypernucleated, detached osteoclasts that are undergoing protracted apoptosis.” And to make matters worse, nobody knows what these strange cells can do!
And last but not least, even though it is a known fact that bisphosphonates can cause osteonecrosis of the jaw, few expected the definitive study in the January 1st issue of Journal of the American Dental Association stating that “even short-term oral use of alendronate led to osteonecrosis of the jaw.” According to Science Daily (January 2, 2009):
This is the first large institutional study in the U.S. to investigate the relationship between oral bisphosphonate use and jaw bone death, said principal investigator Parish Sedghizadeh, assistant professor of clinical dentistry with the USC School of Dentistry. After controlling for referral bias, nine of 208 healthy School of Dentistry patients who take or have taken Fosamax for any length of time were diagnosed with osteonecrosis of the jaw (ONJ). The study’s results are in contrast to drug makers’ prior assertions that bisphosphonate-related ONJ risk is only noticeable with intravenous use of the drugs, not oral usage, Sedghizadeh said. “We’ve been told that the risk with oral bisphosphonates is negligible, but four percent is not negligible,” he said. The results confirm the suspicions of many in the oral health field, he said. “Here at the School of Dentistry we’re getting two or three new patients a week that have bisphosphonate-related ONJ,” he said, “and I know we’re not the only ones seeing it.”
I am really not surprised that this information is now divulged by mainstream medicine because sooner or later, as in the case of Vioxx, the truth will be known. My heart is with all those who are suffering the horrible consequences of being human guinea pigs. As the list of serious side-effects of bisphosphonates continues to grow, I hope that those who are given a prescription will question its value and at least try to discover the truth about these drugs and seek a safe and natural alternative.







Hello, I just came across your page. I am an intestinal failure patient and suffer from SBS, malobsorption, osteoporosis, Crohns disease and psoriatic arthritis. Yesterday I was infused with both Reclast and Remiciad. Today I am sick with a fever of 102, severe lethargy, nausea, chest pain and joint pain and weakness. Help! I feel like a major mistake was made infusing both treatments on the same day.
Try CBDs. I recommend Bluebird Botanicals CBD Hemp oil extract. Classic 6x. Get off the prescribed drugs, they will kill you.
I have systemic lupus in remission.
I used medical marijuana in the form of Rick Simpson Oil to cure the Psoriatic arthritis. It is gone.
I have no signs of lupus.
The CBD/THC treatment is excellent. If you use either or both of those and get off the man-made posions, you will feel better. I promise. 🙂
have been using actonel for 4 months. constant stomach aches, loose stools,
general feeling, nor good. nervous to eat out. have deided to stop using this. have requestd colonoscopy, and contacted a natureapth.
anyone else having the sme problems?
I am completely messed up from one month of Fosamax(generic)! Was unable to sleep,burning stomach, got arthritis pains in fingers and wrists, swelling in knees and elbows. ANXIETY-off the charts. I have been off a month and have severe stiffness in neck and upper back. Also get HORRIBLE neck spasms. Have been back to my Rheumatologist. All blood tests came back normal. He put me on Cymbalta, Aleve and muscle relaxers. None of these are working well yet. I went from a hiking, kayaking,weight lifting 62 year old to a couch residing Momma. Sure hope this pain goes away soon. It’s been a month since I got off the drug. How long till I feel better???
Diane, it’s so sad to hear about the issues you’re experiencing. If you want to get the drugs out of your system faster, I encourage you to check out Rapid Cleanse. It’s a 7-day cleanse designed to rid the body of toxic osteoporosis drugs. You can get the details here:
https://saveourbones.com/rapid-cleanse-sale/
I wish you a speedy recovery!
Dear Vivian,
I sincerely appreciate your program and am grateful that you are warning woman about the dangers of osteoporosis drugs. But I think that your wonderful website is missing a key piece of information. Over 1,000 people in the U.S will develop weak bones because of hyperparathyroidism, a disease in which a parathyroid gland develops a non-cancerous tumor. The tumor secretes too much parathyroid hormone, which causes the bones to release too much calcium into the blood. This results in osteopenia and osteoporosis. Exercise and diet will not cure hyperparathyroidism.
Too few people — doctors included — recognize the link between osteoporosis and hyperparathyroidism. This video by Dr. James Norman shows the bone breakdown process caused by the elevated PTH in hyperparathyroid patients. I would be so grateful if you could let your website visitors know about it. https://youtu.be/sD9st1ZPFrQ
Thank you very kindly,
Jennifer
Can you address the problem of discordance between hip and spine bone density?
Hello,
I am 45 years old and have osteoporosis of the hip and ostopenia of the spine. I have purchased your programme and intend to go along this course rather than medicate. However, my endocrinologist has recommended that I go on the Microgynon oestrogen contracepion pill in order to get through menopause without further bone loss. I have started taking these and just do not feel myself. Have put on weight and am more hungry, moody, lethargic and tired. Has anyone else gone down this road? and I am interested to hear your take on this Vivian? Thanks for everything.
Hi Caroline, I don’t recommend hormone replacement; it’s unnatural to supplement hormones that normally dwindle with age. As mammals, we were created so that we can maintain our health without interfering with the normal biological cycles.
Hi Trudi,
Just go to https://saveourbones.com/snd and click on any of the yellow “Add to Cart” links below the video.
I have Barrett’s Esophagus after taking Simivastatin for 2 yrs. I quit 2 yrs of aldronate/Fosamax 2 yrs ago and a Reclast infusion 1 yr ago. I’m in pain with this dry chronic bumps throat. I quit all Simivastatin, vitamins, citrus, etc n still choke on foods. My older brother just died after finding out he had esophagus cancer that went to lymph and brain and was dead in one month. I’m scared as I still have a 91 mom to take care of and younger brother died of stroke. I’m normally a VERY healthy 60 yr old. Suggestions?
If the problem is brekon auto-responders then try to solve that problem. I was the Director of Support for a good size startup. Our emails always had reply-tos that went into our support system. And I my email account was set up as the catch all . There is room for tools to manage this situation but it’s really not that big of a deal.The thing that is exasperating is that these companies otherwise spend millions of dollars trying to talk to customers and prospective customers. And yet refuse to accept email from them.
I am interested in finding out since I have suffered 3 fractures to the T8 T9 if there is hope for me. I to experienced the scare tactics from my Doctors but choose to tear the prescription up. I am suffering with my hip and my spine. Is there something out there that I can do to stop and reverse since I am already fracturing. I am 56 and the suffering is becoming unbearable. I have not be able to find out from any Doctor what exercise I can safely do it seems like they do not want to talk to you if you do not take their prescription. They actually get mad at me. Please someone help me.
Please tell me your opinion of Evista as a treatment for my osteoporosis of the spine, and osteopenia of both hips. Some articles I have read say that once you have osteoporosis, it can’t be changed but only halted. Is this right? I realize these are two separate questions, but would sincerely appreciate your opinion on both. Thank you
I have stopped my medication Actinol now for one month now! My doctor ordered Forteo for me last month. Instead,I have now ordered from the dent sale “Save our bones program…I believe I can improve my own bones> I am so hopeful to see results by my next bone density tests which I also read are not always accurate…Vivian can you comment or do an article on this info on bone density testing? Thanks Betty
Thanks for the information on osterporosis drugs. I have a dental history so I have decided against the drugs. Brenda
A doctor gave me a free 4 months supply of Actonel a few years ago, when I read the directions on the pkg. I threw it all in the trash, I have gerd, and I will not put that stuff in my stomach. He also gave me a prescription for Vioxx, and I took it for a while, but, since it didn’t help me, I quit its use too. Later I saw the recall. I just live with the bone loss. I am afraid of doctors, anymore. Have had cancer three times. I did have the Chemo treatments the last time, and Retuxin. I am 76 years old.
Hi Vivian
I have been taking Fosamax since 2003. I also take a large dose of calcium every day. A dislocation in my back causes me quite alot of difficulty.I cannot exercise and walk as much as i used to.
I also notice difficulty in swallowing, and have to drink alot of water to get my food down!
Is it safe to STOP Alendronate at this stage?
SUJATHA
I agree with you re: the potentially negative effects of these pharmaceuticals. However, I would also like to hear your perspective on Forteo which is a different approach. It is a teriparatide developed & marketed by Lily.
My wife has (had) an osteoporosis condition so severe that a minor backyard fall shattered her L1 & did other major damage resulting in 3 surgeries in just a 9 day period. Post surgery her last Dex was 50% WORSE than the previous 6 month scan even tho she has been on mega doses of Calcium carbonate (I now know this is not the “GOOD” calcium) and substantial doses of D3.
She is in such chronic pain & fear of additional fractures that she is bedridden 50% of the time. Please tell me what you know of the functional mechanism & methodology of the Forteo medication, as we are currently trying to evaluate it. She has been taking it for a couple months now. Thank you very much for your work, Viv
Max Bennett